
What are the three main varibles of the gas laws – What are the three main variables of the gas laws sets the stage for this enthralling narrative, offering readers a glimpse into a story that is rich in detail and brimming with originality from the outset. The study of gas laws is a fascinating journey into the world of invisible particles and their predictable behavior. Understanding these laws allows us to unravel the mysteries behind how gases respond to changes in pressure, volume, and temperature. Imagine a balloon filled with air, what happens when you squeeze it? Or when you heat it up? The answers lie within the realm of gas laws, and we are about to embark on an exploration of these fundamental principles.
The three main variables that govern the behavior of gases are pressure, volume, and temperature. Pressure is the force exerted by gas molecules on the walls of their container. Volume refers to the space occupied by the gas. And temperature is a measure of the average kinetic energy of the gas molecules. These variables are interconnected, and changes in one can significantly affect the others. The gas laws, discovered through meticulous experimentation and observation, provide us with the mathematical relationships that describe these interactions.
Gay-Lussac’s Law
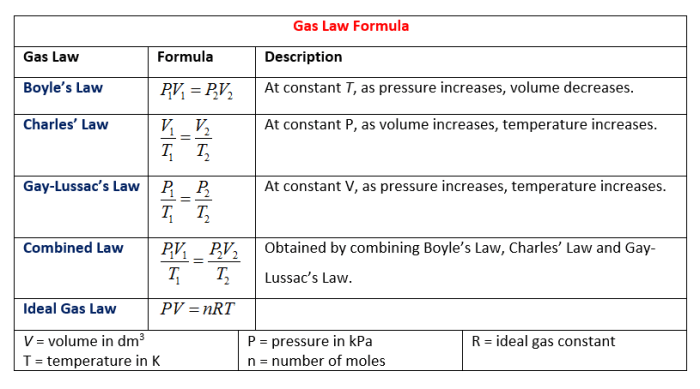
Gay-Lussac’s Law, also known as the pressure-temperature law, describes the relationship between the pressure and temperature of a gas when the volume is held constant. This law is a fundamental principle in chemistry and physics, explaining the behavior of gases under various conditions.
Mathematical Expression of Gay-Lussac’s Law
Gay-Lussac’s Law states that the pressure of a fixed mass of gas is directly proportional to its absolute temperature when the volume is held constant. This relationship can be expressed mathematically as:
P1/T1 = P2/T2
Where:
* P1 is the initial pressure
* T1 is the initial absolute temperature
* P2 is the final pressure
* T2 is the final absolute temperature
Direct Relationship Between Pressure and Temperature
Gay-Lussac’s Law highlights a direct relationship between the pressure and temperature of a gas. This means that as the temperature of a gas increases, its pressure also increases proportionally, assuming the volume remains constant. Conversely, if the temperature decreases, the pressure will decrease proportionally.
Real-World Applications of Gay-Lussac’s Law
Gay-Lussac’s Law has numerous real-world applications, including:
* Pressure Cookers: Pressure cookers operate on the principle of Gay-Lussac’s Law. By increasing the temperature inside the cooker, the pressure also increases, allowing food to cook faster.
* Hot Air Balloons: Hot air balloons rise because the heated air inside the balloon expands, decreasing its density and creating buoyancy. This expansion of air is governed by Gay-Lussac’s Law.
* Tire Pressure: As the temperature of a tire increases, the pressure inside the tire also increases. This is why it is important to check tire pressure regularly, especially during hot weather.
* Aerosol Cans: Aerosol cans contain pressurized gases that are released when the can is sprayed. The pressure inside the can is directly proportional to the temperature, which is why it is important to avoid exposing aerosol cans to extreme temperatures.
Combined Gas Law
The Combined Gas Law is a fundamental principle in chemistry that combines Boyle’s Law, Charles’s Law, and Gay-Lussac’s Law to describe the relationship between the pressure, volume, and temperature of an ideal gas. It provides a comprehensive framework for understanding how these variables interact under different conditions.
The Combined Gas Law states that the ratio of the product of pressure and volume to the absolute temperature of a fixed amount of gas is a constant. This can be expressed mathematically as:
P₁V₁/T₁ = P₂V₂/T₂
Where:
* P₁ is the initial pressure
* V₁ is the initial volume
* T₁ is the initial absolute temperature
* P₂ is the final pressure
* V₂ is the final volume
* T₂ is the final absolute temperature
Combining Gas Laws, What are the three main varibles of the gas laws
The Combined Gas Law effectively combines the three individual gas laws:
* Boyle’s Law: This law states that the volume of a gas is inversely proportional to its pressure at constant temperature.
* Charles’s Law: This law states that the volume of a gas is directly proportional to its absolute temperature at constant pressure.
* Gay-Lussac’s Law: This law states that the pressure of a gas is directly proportional to its absolute temperature at constant volume.
The Combined Gas Law encompasses these relationships by considering the combined effect of all three variables. It allows us to predict how the pressure, volume, and temperature of a gas will change when one or more of these variables are altered, assuming the amount of gas remains constant.
Applications of the Combined Gas Law
The Combined Gas Law has numerous applications in various fields, including:
* Chemistry: Determining the volume of a gas at a different temperature or pressure.
* Meteorology: Predicting weather patterns and atmospheric conditions.
* Engineering: Designing and operating systems involving gases, such as engines and gas pipelines.
* Aerospace: Calculating the behavior of gases in aircraft and spacecraft.
Here are some examples of how the Combined Gas Law can be used to solve problems:
* Example 1: A balloon filled with helium has a volume of 2.0 L at a temperature of 25°C and a pressure of 1.0 atm. What will be the volume of the balloon if the temperature is increased to 50°C and the pressure is reduced to 0.5 atm?
To solve this, we can use the Combined Gas Law equation:
P₁V₁/T₁ = P₂V₂/T₂
Substituting the given values:
(1.0 atm)(2.0 L)/(25°C + 273.15) = (0.5 atm)(V₂)/(50°C + 273.15)
Solving for V₂, we get:
V₂ = 4.0 L
Therefore, the volume of the balloon will increase to 4.0 L.
* Example 2: A scuba diver is at a depth of 30 meters where the pressure is 4.0 atm. The diver’s air tank has a volume of 10 L at the surface where the pressure is 1.0 atm. What will be the volume of the air in the tank at the depth of 30 meters?
Using the Combined Gas Law equation:
P₁V₁/T₁ = P₂V₂/T₂
Assuming the temperature remains constant, we can simplify the equation to:
P₁V₁ = P₂V₂
Substituting the given values:
(1.0 atm)(10 L) = (4.0 atm)(V₂)
Solving for V₂, we get:
V₂ = 2.5 L
Therefore, the volume of the air in the tank will decrease to 2.5 L at the depth of 30 meters.
Ideal Gas Law

The Ideal Gas Law is a fundamental law in chemistry that describes the behavior of ideal gases. It is a combination of the previously discussed gas laws, including Boyle’s Law, Charles’s Law, Gay-Lussac’s Law, and Avogadro’s Law. This law provides a simple and accurate way to predict the behavior of gases under various conditions.
The Ideal Gas Law expresses the relationship between pressure (P), volume (V), temperature (T), and the number of moles (n) of an ideal gas. It states that the product of pressure and volume is directly proportional to the product of the number of moles and the absolute temperature.
Mathematical Expression
The Ideal Gas Law is mathematically expressed as:
PV = nRT
where:
– P is the pressure of the gas in atmospheres (atm)
– V is the volume of the gas in liters (L)
– n is the number of moles of gas
– R is the ideal gas constant, which is equal to 0.0821 L atm/mol K
– T is the temperature of the gas in Kelvin (K)
Relationship Between Pressure, Volume, Temperature, and Number of Moles
The Ideal Gas Law demonstrates the following relationships:
– Pressure and Volume: Pressure and volume are inversely proportional. This means that if the pressure of a gas increases, the volume will decrease, and vice versa, assuming the temperature and number of moles remain constant.
– Temperature and Volume: Temperature and volume are directly proportional. As the temperature increases, the volume will also increase, and vice versa, assuming pressure and the number of moles remain constant.
– Temperature and Pressure: Temperature and pressure are directly proportional. As the temperature increases, the pressure will also increase, and vice versa, assuming volume and the number of moles remain constant.
– Number of Moles and Volume: The number of moles and volume are directly proportional. As the number of moles increases, the volume will also increase, and vice versa, assuming pressure and temperature remain constant.
Limitations of the Ideal Gas Law
The Ideal Gas Law is a theoretical model that assumes ideal gas behavior. In reality, gases deviate from ideal behavior under certain conditions, such as high pressure or low temperature.
– High Pressure: At high pressures, the gas molecules are closer together, and their intermolecular forces become more significant. This leads to deviations from the ideal gas law, as the actual volume occupied by the molecules is no longer negligible compared to the total volume.
– Low Temperature: At low temperatures, the gas molecules move slower, and their intermolecular forces become more dominant. This can also cause deviations from the ideal gas law.
– Real Gases: Real gases have intermolecular forces and finite molecular volumes, which are not considered in the ideal gas law. Therefore, the Ideal Gas Law is an approximation and may not accurately represent real gas behavior under all conditions.
Applications of Gas Laws: What Are The Three Main Varibles Of The Gas Laws

The gas laws are not merely theoretical concepts; they have profound implications for various aspects of our lives. From weather forecasting to automotive engineering, these laws play a crucial role in understanding and predicting the behavior of gases in different scenarios.
Real-World Applications of Gas Laws
The gas laws have numerous applications in various fields, impacting our daily lives in ways we may not even realize. Here is a table showcasing some key applications:
| Gas Law | Application | Description | Example |
|---|---|---|---|
| Boyle’s Law | Weather Forecasting | Meteorologists use Boyle’s Law to understand how atmospheric pressure changes with altitude, influencing weather patterns. | As air rises, the pressure decreases, causing air to expand, cool, and potentially form clouds or precipitation. |
| Charles’s Law | Automotive Engines | Charles’s Law is crucial in the design and operation of internal combustion engines. It explains how the volume of air in the cylinder expands when heated by combustion, driving the piston. | The temperature increase during combustion causes the air volume to expand, creating pressure that pushes the piston down, generating power. |
| Gay-Lussac’s Law | Diving and Scuba Equipment | Divers rely on Gay-Lussac’s Law to understand how the pressure of air in scuba tanks changes with depth. This law is essential for ensuring safe diving practices. | As a diver descends, the pressure increases, compressing the air in the tank. Divers need to be aware of this pressure change to avoid exceeding safe limits. |
| Combined Gas Law | Aerospace Engineering | Aerospace engineers use the Combined Gas Law to analyze the behavior of gases in high-altitude environments, where pressure and temperature fluctuate significantly. | The Combined Gas Law helps engineers design aircraft systems that can operate effectively under varying atmospheric conditions. |
| Ideal Gas Law | Chemical Reactions and Processes | The Ideal Gas Law is fundamental to understanding chemical reactions involving gases. It allows scientists to predict the behavior of gases under various conditions, such as temperature, pressure, and volume. | The Ideal Gas Law is used to calculate the yield of a chemical reaction involving gases, ensuring efficient and safe production processes. |
Final Summary
The exploration of gas laws has unveiled the intricate dance between pressure, volume, and temperature. These fundamental principles, derived from meticulous observation and experimentation, offer a profound understanding of the behavior of gases. From weather forecasting to the design of rocket engines, gas laws find applications across diverse scientific and engineering fields. As we delve deeper into the world of gases, we discover the profound implications of these laws, reminding us that even the seemingly invisible can be governed by predictable and elegant rules.
FAQ Section
What is the difference between Boyle’s Law and Charles’s Law?
Boyle’s Law describes the inverse relationship between pressure and volume at constant temperature, while Charles’s Law describes the direct relationship between volume and temperature at constant pressure.
How do gas laws relate to real-world phenomena?
Gas laws have numerous applications in various fields, including weather forecasting, automotive engines, diving, aerospace engineering, and chemical reactions.
What are some limitations of the Ideal Gas Law?
The Ideal Gas Law assumes that gas molecules have no volume and do not interact with each other, which is not entirely accurate for real gases, especially at high pressures and low temperatures.




