
What does the law of conservation of mass state? This fundamental principle of chemistry and physics asserts that in an isolated system, the total mass of the reactants before a chemical reaction must equal the total mass of the products after the reaction. Imagine a simple experiment where you mix baking soda and vinegar – the resulting fizz and bubbling might make you think something has disappeared, but the law of conservation of mass tells us that the total mass of the mixture remains constant, even though it transforms into different substances. This principle, discovered by Antoine Lavoisier in the 18th century, is a cornerstone of our understanding of matter and its transformations.
The law of conservation of mass has far-reaching implications, shaping our understanding of chemical reactions, engineering designs, and even environmental science. It plays a crucial role in understanding how matter is transformed and recycled in nature, helping us to develop sustainable practices. The law also underpins many technological advancements, from the production of fuels to the development of new materials.
The Law of Conservation of Mass

The Law of Conservation of Mass is a fundamental principle in chemistry and physics that states that in an isolated system, the total mass of the reactants before a chemical reaction must equal the total mass of the products after the reaction. This means that matter cannot be created or destroyed, only transformed from one form to another.
This principle has significant implications for understanding chemical reactions, as it allows us to predict the amount of reactants and products involved in a given reaction. It also forms the basis for many other scientific laws and theories, such as the law of definite proportions and the law of multiple proportions.
Historical Context of the Law’s Discovery
The concept of the conservation of mass was first proposed by ancient Greek philosophers, but it was Antoine Lavoisier, a French chemist, who is credited with experimentally proving the law in the late 18th century. Lavoisier conducted numerous experiments with carefully measured reactants and products, and he found that the total mass of the reactants always equaled the total mass of the products. This discovery was a major breakthrough in the understanding of chemical reactions, and it laid the foundation for modern chemistry.
Examples of Everyday Phenomena Demonstrating the Law of Conservation of Mass
The law of conservation of mass is demonstrated in many everyday phenomena. For example, when you burn a piece of wood, the wood appears to disappear, but it has actually been transformed into ash, smoke, and gases. The total mass of the ash, smoke, and gases will equal the mass of the original piece of wood.
Another example is when you mix baking soda and vinegar. The two substances react to form carbon dioxide gas, which is released into the air. The total mass of the baking soda, vinegar, and carbon dioxide gas will equal the mass of the original baking soda and vinegar.
Key Concepts and Definitions
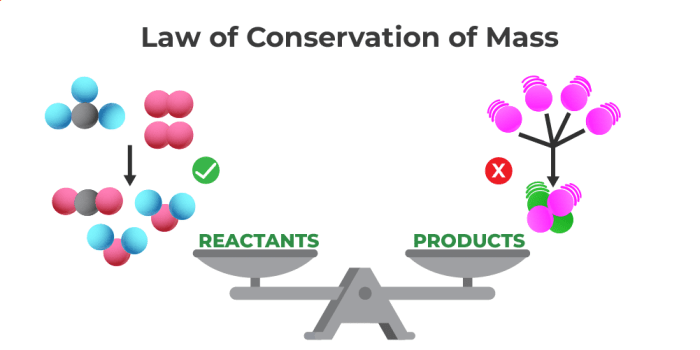
To fully understand the Law of Conservation of Mass, it is crucial to grasp the key concepts and definitions that underpin it.
Mass and Matter
Mass is a fundamental property of matter, representing the amount of matter an object contains. Matter is anything that occupies space and has mass. It exists in various states, including solid, liquid, and gas.
Mass vs. Weight
While often used interchangeably, mass and weight are distinct concepts.
- Mass is a measure of the amount of matter in an object, and it remains constant regardless of location. It is a scalar quantity, meaning it only has magnitude.
- Weight, on the other hand, is the force exerted on an object due to gravity. It is a vector quantity, having both magnitude and direction.
For example, a person’s mass remains the same on Earth and the Moon. However, their weight differs because the gravitational force on the Moon is weaker than on Earth.
Closed System
The Law of Conservation of Mass applies to a closed system, which is a system where no matter can enter or leave. This means that the total mass within the system remains constant, even if chemical or physical changes occur within it.
A closed system is crucial for the Law of Conservation of Mass to hold true.
Applications of the Law of Conservation of Mass
The law of conservation of mass is a fundamental principle in chemistry and physics that has wide-ranging applications across various fields. It is a cornerstone for understanding how matter behaves in chemical reactions and physical processes.
Chemical Reactions
The law of conservation of mass is crucial for understanding chemical reactions. It dictates that in any closed system, the total mass of the reactants before a chemical reaction must equal the total mass of the products after the reaction. This principle is reflected in balanced chemical equations, which represent the quantitative relationships between reactants and products.
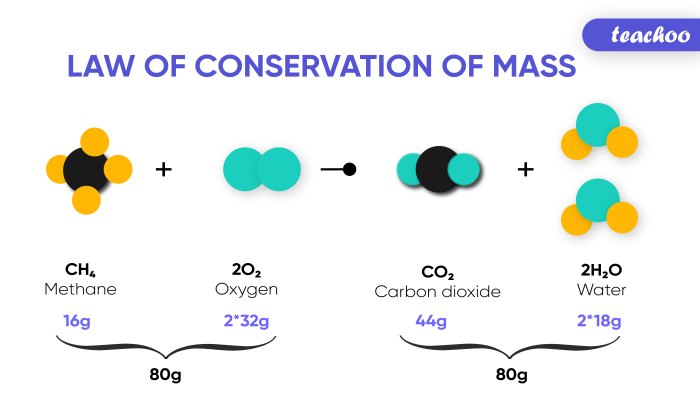
For example, in the combustion of methane (CH4) with oxygen (O2) to produce carbon dioxide (CO2) and water (H2O), the balanced chemical equation is:
CH4 + 2O2 → CO2 + 2H2O
This equation indicates that one molecule of methane reacts with two molecules of oxygen to produce one molecule of carbon dioxide and two molecules of water. The total number of atoms of each element on the reactant side (1 carbon, 4 hydrogen, and 4 oxygen) is equal to the total number of atoms of each element on the product side. This ensures that mass is conserved throughout the reaction.
Demonstration of the Law of Conservation of Mass
A simple experiment can demonstrate the law of conservation of mass.
1. Materials: A sealed container (e.g., a flask or a jar), a balance scale, a small amount of baking soda (sodium bicarbonate), vinegar (acetic acid), and a balloon.
2. Procedure:
* Weigh the sealed container with the baking soda inside.
* Attach the balloon to the container’s opening.
* Pour vinegar into the container through the balloon’s opening.
* Observe the reaction between baking soda and vinegar, which produces carbon dioxide gas that inflates the balloon.
* After the reaction is complete, weigh the sealed container again.
3. Observation: You will notice that the total mass of the container and its contents remains the same before and after the reaction, even though the balloon is inflated with carbon dioxide gas.
This experiment illustrates that even though a chemical reaction takes place, the total mass of the system remains constant. The mass of the carbon dioxide gas produced is accounted for by the decrease in the mass of the reactants (baking soda and vinegar).
Applications in Various Fields
The law of conservation of mass has wide-ranging applications in various fields:
- Engineering: Engineers use this law in designing and analyzing processes involving chemical reactions, such as combustion in power plants and chemical synthesis in manufacturing industries. They ensure that the mass of reactants and products is accounted for to optimize efficiency and minimize waste.
- Environmental Science: The law is crucial in understanding environmental processes, such as the cycling of elements (e.g., carbon, nitrogen) in ecosystems. It helps assess the impact of human activities on the environment, including pollution and climate change.
- Medicine: The law of conservation of mass plays a role in understanding metabolic processes in the human body. It helps explain how nutrients are broken down and transformed into energy, and how waste products are eliminated. It is also applied in medical imaging techniques, such as positron emission tomography (PET scans), which rely on the principle of radioactive decay and mass conservation.
Limitations and Exceptions: What Does The Law Of Conservation Of Mass State
While the law of conservation of mass holds true in many everyday chemical reactions, it has limitations, particularly in the realm of nuclear reactions and when considering the relationship between mass and energy.
Nuclear Reactions
Nuclear reactions involve changes at the atomic level, where the nuclei of atoms are transformed. In these reactions, the total mass of the reactants is not always equal to the total mass of the products. This discrepancy arises because some of the mass is converted into energy or vice versa, as described by Einstein’s famous equation, E=mc².
E=mc²
This equation states that energy (E) is equivalent to mass (m) multiplied by the speed of light squared (c²). It implies that mass and energy are interchangeable, and a small amount of mass can be converted into a significant amount of energy, or vice versa.
Mass-Energy Equivalence
In nuclear reactions, the difference in mass between reactants and products is often referred to as the “mass defect.” This mass defect is directly proportional to the energy released or absorbed in the reaction. For instance, in nuclear fission, a heavy nucleus splits into lighter nuclei, releasing a tremendous amount of energy. This energy release comes at the expense of a small decrease in mass. Conversely, in nuclear fusion, lighter nuclei combine to form a heavier nucleus, releasing energy and a small decrease in mass.
Examples of Scenarios
- Nuclear Fission: In the fission of uranium-235, a neutron strikes the uranium nucleus, causing it to split into two lighter nuclei, such as barium and krypton, along with several neutrons. The total mass of the products is slightly less than the mass of the original uranium nucleus, with the missing mass converted into energy, primarily in the form of kinetic energy of the fission products and emitted neutrons.
- Nuclear Fusion: In the fusion of hydrogen isotopes, such as deuterium and tritium, to form helium, a significant amount of energy is released. The mass of the helium nucleus is slightly less than the combined mass of the deuterium and tritium nuclei. This mass difference is converted into energy, contributing to the immense energy output of the Sun and other stars.
- Radioactive Decay: Radioactive isotopes undergo spontaneous decay, emitting particles and energy. The mass of the daughter nucleus is less than the mass of the parent nucleus. This mass difference is released as energy in the form of radiation.
Significance and Impact
The Law of Conservation of Mass is a fundamental principle in science, impacting numerous fields and influencing our understanding of the universe. Its significance extends beyond theoretical concepts, influencing practical applications and technological advancements.
The law’s impact can be seen in its role in the development of modern chemistry and physics. It provides a framework for understanding chemical reactions and the behavior of matter, allowing scientists to predict and explain phenomena.
Importance in Various Fields, What does the law of conservation of mass state
The Law of Conservation of Mass plays a crucial role in various fields, including:
- Chemistry: The law is fundamental to stoichiometry, the study of quantitative relationships between reactants and products in chemical reactions. It allows chemists to predict the amount of reactants and products involved in a reaction, enabling accurate calculations and efficient chemical processes.
- Physics: In physics, the law is essential for understanding energy conservation, as mass and energy are interconnected through Einstein’s famous equation E=mc². This principle is vital for understanding nuclear reactions and the behavior of particles at the atomic level.
- Engineering: Engineers apply the law in various disciplines, including chemical engineering, mechanical engineering, and environmental engineering. For example, in chemical engineering, the law helps in designing reactors and optimizing processes, while in environmental engineering, it aids in understanding and managing waste disposal and pollution control.
- Environmental Science: The law is crucial for understanding the cycling of elements in the environment. It helps in analyzing the movement of pollutants and understanding the impact of human activities on the environment.
- Medicine: The law plays a role in medical imaging techniques, such as PET scans and MRI, where the distribution of radioactive isotopes or magnetic fields is used to diagnose diseases.
Role in the Development of Modern Chemistry and Physics
The Law of Conservation of Mass was a crucial stepping stone in the development of modern chemistry and physics. It provided a foundation for the development of atomic theory and the understanding of chemical reactions.
“The total mass of a closed system remains constant over time, regardless of changes within the system.” – Antoine Lavoisier
Lavoisier’s work, based on the Law of Conservation of Mass, revolutionized chemistry by demonstrating that matter is not lost or gained during chemical reactions. This led to the development of the concept of atoms and molecules and the understanding of chemical reactions as rearrangements of atoms.
In physics, the law was integrated into the theory of relativity, which established the relationship between mass and energy. This led to the development of nuclear physics and the understanding of nuclear reactions, such as nuclear fission and fusion.
Influence on Scientific Understanding and Technological Advancements
The Law of Conservation of Mass has significantly influenced scientific understanding and technological advancements.
- Chemical Synthesis: The law is crucial for chemical synthesis, allowing scientists to predict the yield of reactions and optimize processes for producing desired products.
- Pollution Control: The law helps in understanding the fate of pollutants in the environment, allowing for the development of strategies for pollution control and environmental remediation.
- Nuclear Energy: The law is fundamental to the development of nuclear energy, as it governs the conversion of mass into energy in nuclear reactions.
- Space Exploration: The law is essential for understanding the behavior of spacecraft and the propulsion systems used for space travel.
Ultimate Conclusion
The law of conservation of mass, though seemingly simple, has profound implications for our understanding of the universe. It serves as a bedrock principle in chemistry and physics, guiding our exploration of matter and its transformations. From the everyday phenomenon of cooking to the complexities of nuclear reactions, the law of conservation of mass provides a framework for comprehending the fundamental processes that govern our world. As we continue to delve deeper into the mysteries of the universe, the law of conservation of mass will undoubtedly continue to play a vital role in shaping our scientific understanding.
FAQ Summary
Does the law of conservation of mass apply to all reactions?
While the law of conservation of mass is generally true, it does have some limitations. For example, it doesn’t hold true in nuclear reactions where mass can be converted into energy and vice versa, as described by Einstein’s famous equation E=mc².
What are some examples of the law of conservation of mass in everyday life?
Burning wood is a great example. While the wood appears to disappear, it’s actually transformed into ash, smoke, and gases. The total mass of these products equals the initial mass of the wood. Another example is baking a cake. The ingredients you mix together have a combined mass equal to the mass of the finished cake, even though it looks different.
Why is the law of conservation of mass important in environmental science?
The law helps us understand how pollutants are transformed and spread in the environment. It also guides the development of methods for waste management and pollution control. For example, understanding how pollutants are transformed in the atmosphere helps us develop strategies to reduce air pollution.




