What is Bohr’s Law? It’s a question that delves into the fascinating world of atomic structure, a world where electrons dance around the nucleus in specific orbits, their energy levels governed by a set of rules. Niels Bohr, a brilliant Danish physicist, revolutionized our understanding of the atom with his groundbreaking model, proposing that electrons could only exist in specific energy levels, like rungs on a ladder. This model, now known as Bohr’s Law, laid the foundation for modern quantum mechanics and continues to be a cornerstone of our understanding of the atom.
Bohr’s Law challenged the prevailing scientific thought, which saw the atom as a miniature solar system with electrons orbiting the nucleus in any possible path. Bohr’s model introduced the concept of quantized energy levels, meaning that electrons could only occupy specific energy states, and transitions between these states were accompanied by the absorption or emission of photons, the fundamental particles of light. This revolutionary idea explained many observed phenomena, such as the spectral lines emitted by excited atoms, which had previously been a mystery.
Niels Bohr
Niels Bohr, a renowned physicist from Denmark, made groundbreaking contributions to the understanding of atomic structure and quantum mechanics. His life and career were marked by a deep intellectual curiosity and a commitment to scientific exploration.
Early Life and Education
Born in Copenhagen in 1885, Niels Bohr received his early education at the Gammelholm Latin School. His father, Christian Bohr, was a renowned physiologist, and his mother, Ellen Adler Bohr, came from a wealthy Jewish family. Bohr’s academic brilliance was evident from a young age, and he excelled in mathematics and physics. He enrolled at the University of Copenhagen in 1903, where he pursued a degree in physics.
Influences on Bohr’s Scientific Journey
Bohr’s scientific journey was profoundly influenced by several key figures. He was particularly inspired by the work of J.J. Thomson, who discovered the electron, and Ernest Rutherford, who proposed the nuclear model of the atom. Bohr also benefited from the intellectual environment at the University of Copenhagen, where he was surrounded by brilliant minds.
Bohr’s Research on Atomic Structure, What is bohr’s law
Bohr’s research on atomic structure revolutionized our understanding of the atom. In 1913, he proposed the Bohr model, which described the atom as a small, positively charged nucleus surrounded by negatively charged electrons orbiting in quantized energy levels. This model successfully explained the line spectra of hydrogen and other elements, a phenomenon that could not be explained by classical physics.
The Bohr Model
The Bohr model, based on the work of Max Planck and Albert Einstein, introduced the concept of quantized energy levels. According to this model, electrons can only occupy specific energy levels, and they can jump between these levels by absorbing or emitting photons of light. The energy of the photon is equal to the difference in energy between the two levels. This model provided a foundation for understanding the stability of atoms and the nature of light emission and absorption.
“An electron can only exist in certain discrete orbits around the nucleus, each with a specific energy level.”
Bohr’s Legacy
Niels Bohr’s work had a profound impact on the development of quantum mechanics and our understanding of the atom. He was awarded the Nobel Prize in Physics in 1922 for his contributions to the understanding of atomic structure. Bohr’s legacy extends beyond his scientific achievements. He was a passionate advocate for international scientific collaboration and played a significant role in the development of the atomic bomb during World War II.
Bohr’s Atomic Model: What Is Bohr’s Law
Bohr’s atomic model, proposed in 1913, revolutionized our understanding of the atom. It was a significant departure from previous models and provided a framework for explaining the observed spectral lines of hydrogen.
Postulates of Bohr’s Atomic Model
Bohr’s model is based on several key postulates that describe the behavior of electrons within an atom:
- Electrons occupy specific energy levels: Electrons in an atom can only exist in specific, discrete energy levels, often called orbits or shells. These energy levels are quantized, meaning they can only have specific, fixed values. This contrasts with classical physics, which allows electrons to have any energy value.
- Electrons transition between energy levels by absorbing or emitting photons: When an electron jumps from a lower energy level to a higher one, it absorbs a photon of light with an energy equal to the difference between the two levels. Conversely, when an electron drops from a higher energy level to a lower one, it emits a photon of light with an energy equal to the difference between the two levels. This explains the observed spectral lines of hydrogen.
- Angular momentum is quantized: The angular momentum of an electron in an orbit is quantized, meaning it can only take on specific values. This postulate is closely related to the quantization of energy levels.
Differences from Rutherford’s Model
Bohr’s model addressed some of the shortcomings of Rutherford’s model, which proposed a planetary model of the atom with a positively charged nucleus and negatively charged electrons orbiting around it. However, Rutherford’s model failed to explain the stability of atoms and the discrete spectral lines observed in atomic emission spectra.
- Stability of atoms: According to classical physics, an orbiting electron should continuously radiate energy and spiral into the nucleus. However, Bohr’s model addressed this issue by postulating that electrons occupy specific energy levels and do not radiate energy when they are in these levels.
- Discrete spectral lines: Rutherford’s model could not explain the discrete spectral lines observed in atomic emission spectra. Bohr’s model explained this phenomenon by proposing that electrons can only transition between specific energy levels, leading to the emission of photons with specific energies and corresponding wavelengths.
Diagram of Bohr’s Model
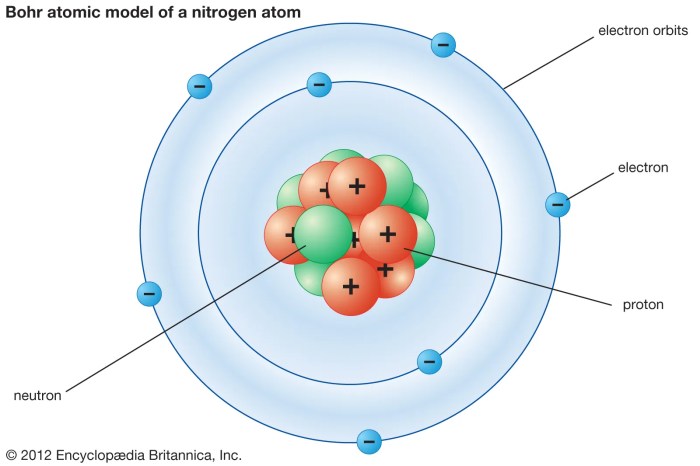
Bohr’s model is often represented with a diagram showing the electron orbits and energy levels. The nucleus is at the center, and the electrons are shown orbiting around it in specific circular paths, called orbits or shells. The orbits are labeled with numbers (n = 1, 2, 3, etc.), representing the energy levels. The lowest energy level (n = 1) is closest to the nucleus, and the higher energy levels are farther away.
Electrons can only exist in specific energy levels (n = 1, 2, 3, etc.), and they can jump between these levels by absorbing or emitting photons of light.
Successes and Limitations of Bohr’s Model
Bohr’s model, despite its simplicity, was remarkably successful in explaining some fundamental aspects of atomic behavior. However, it also had its limitations, paving the way for more sophisticated quantum mechanical models.
Successes of Bohr’s Model
Bohr’s model was a significant step forward in understanding the structure of the atom. It successfully explained several key phenomena, including:
- Atomic Spectra: Bohr’s model successfully explained the observed line spectra of hydrogen atoms. It predicted that electrons could only occupy specific energy levels, and transitions between these levels would result in the emission or absorption of light at specific wavelengths. This was consistent with the experimental observations of atomic spectra.
- Stability of Atoms: Bohr’s model explained the stability of atoms by proposing that electrons could only exist in specific orbits around the nucleus. These orbits were quantized, meaning that electrons could not occupy positions between these allowed orbits. This prevented electrons from spiraling into the nucleus, which classical physics predicted would happen due to the electrostatic attraction between the electron and the nucleus.
Limitations of Bohr’s Model
While Bohr’s model was successful in explaining some atomic phenomena, it had several limitations:
- Fine Structure of Spectral Lines: Bohr’s model could not explain the fine structure of spectral lines, which is the splitting of spectral lines into multiple closely spaced lines. This splitting arises from the interaction of the electron’s spin and orbital angular momentum, a phenomenon not accounted for in Bohr’s model.
- Spectra of Multi-Electron Atoms: Bohr’s model was only successful in explaining the spectra of hydrogen, which has only one electron. It failed to accurately predict the spectra of atoms with multiple electrons. This is because the interactions between multiple electrons are complex and not adequately accounted for in Bohr’s simple model.
- Zeeman Effect: The Zeeman effect, the splitting of spectral lines in the presence of a magnetic field, could not be explained by Bohr’s model. This effect arises from the interaction of the electron’s magnetic moment with the external magnetic field, a phenomenon that was not included in Bohr’s model.
Comparison with Quantum Mechanical Models
Later quantum mechanical models of the atom, such as the Schrödinger model, provided a more accurate and complete description of atomic structure and behavior. These models overcome the limitations of Bohr’s model by incorporating the wave-particle duality of matter and the concept of quantum operators.
- Similarities: Both Bohr’s model and quantum mechanical models recognize that electrons occupy specific energy levels in atoms. However, quantum mechanical models go beyond Bohr’s model by describing these energy levels using wave functions and quantum operators.
- Differences: Bohr’s model assumes that electrons orbit the nucleus in well-defined circular paths. Quantum mechanical models, on the other hand, describe electrons as probability distributions, where the probability of finding an electron at a particular location is given by the square of the wave function.
Bohr’s Legacy and Impact
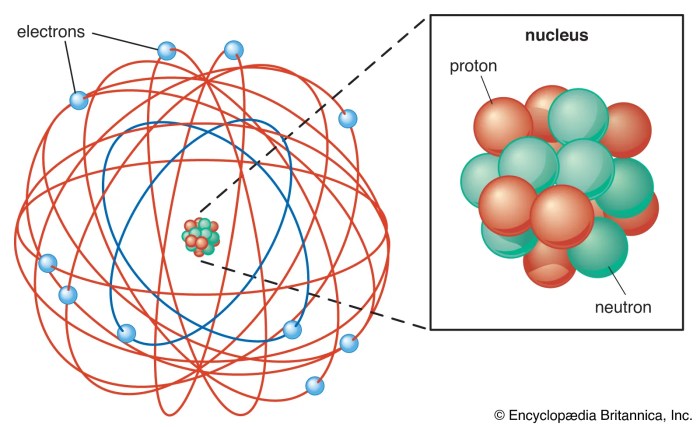
Niels Bohr’s groundbreaking work on the atom laid the foundation for modern physics and continues to influence scientific understanding today. His contributions were instrumental in shaping our understanding of the fundamental building blocks of matter and the nature of light.
Influence on Quantum Mechanics
Bohr’s atomic model, while ultimately superseded by more sophisticated theories, provided a crucial stepping stone towards the development of quantum mechanics. His postulates, such as the quantization of energy levels and the role of electrons in atomic transitions, challenged classical physics and paved the way for a new understanding of the atom. His work, alongside that of other pioneers like Max Planck and Albert Einstein, contributed to the birth of quantum mechanics, which revolutionized our understanding of the universe at its most fundamental level.
Impact on Understanding Atomic Structure
Bohr’s model offered a revolutionary explanation for the stability of atoms and the emission of specific wavelengths of light, known as spectral lines. He proposed that electrons occupy quantized energy levels, and transitions between these levels correspond to the absorption or emission of photons with specific energies. This model provided a framework for understanding the structure of atoms and their interactions with light. It also laid the groundwork for future advancements in understanding chemical bonding, atomic spectroscopy, and other fields.
The Institute for Theoretical Physics
In 1920, Bohr established the Institute for Theoretical Physics in Copenhagen, which became a renowned hub for scientific innovation and collaboration. The institute attracted some of the brightest minds in physics, including Werner Heisenberg, Wolfgang Pauli, and Paul Dirac, who contributed significantly to the development of quantum mechanics. The institute fostered an environment of intellectual exchange and critical thinking, leading to groundbreaking discoveries and advancements in theoretical physics. Bohr’s leadership and vision transformed the institute into a global center for scientific excellence, solidifying his legacy as a visionary leader in the scientific community.
Summary
Bohr’s Law, though not without its limitations, was a pivotal moment in the development of quantum mechanics. It provided a framework for understanding the behavior of electrons in atoms, paving the way for more sophisticated models that would later emerge. Today, Bohr’s Law continues to be a valuable tool for understanding the fundamentals of atomic structure, serving as a stepping stone to the more complex world of quantum mechanics. It reminds us that even seemingly simple questions, like “what is Bohr’s Law?”, can lead us down fascinating paths of scientific discovery.
FAQ
What are the key postulates of Bohr’s atomic model?
Bohr’s model postulates that electrons can only exist in specific energy levels, called orbits, around the nucleus. These orbits are quantized, meaning that electrons can only jump between these specific levels, absorbing or emitting photons in the process. The energy of the emitted or absorbed photon corresponds to the difference in energy between the two orbits.
How does Bohr’s model differ from Rutherford’s model?
Rutherford’s model, based on his gold foil experiment, proposed that atoms have a dense, positively charged nucleus surrounded by negatively charged electrons orbiting in any possible path. Bohr’s model, however, introduced the concept of quantized energy levels, stating that electrons can only exist in specific orbits, and transitions between these orbits involve the absorption or emission of photons.
What are the limitations of Bohr’s model?
Bohr’s model was successful in explaining the spectral lines of hydrogen, but it failed to account for the fine structure of spectral lines observed in other elements. It also couldn’t explain the splitting of spectral lines in the presence of a magnetic field (Zeeman effect). Moreover, Bohr’s model was unable to predict the probabilities of finding electrons in different locations around the nucleus, a concept that was later explained by quantum mechanics.