
What is k in coulomb’s law – What is ‘k’ in Coulomb’s Law? This constant, often referred to as Coulomb’s constant, plays a crucial role in understanding the fundamental force of attraction or repulsion between electrically charged objects. Coulomb’s Law, a cornerstone of electrostatics, quantifies this force, and ‘k’ acts as a proportionality factor that helps determine the strength of the interaction.
Imagine two charged objects, one positively charged and the other negatively charged. They exert a force on each other, either pulling them closer (attraction) or pushing them apart (repulsion). The magnitude of this force depends on several factors, including the charges themselves and the distance separating them. Coulomb’s Law, expressed mathematically as F = k * (q1 * q2) / r², captures this relationship, where ‘k’ is the proportionality constant, ‘q1’ and ‘q2’ are the magnitudes of the charges, and ‘r’ is the distance between them. This equation tells us that the force is directly proportional to the product of the charges and inversely proportional to the square of the distance.
Applications of Coulomb’s Law
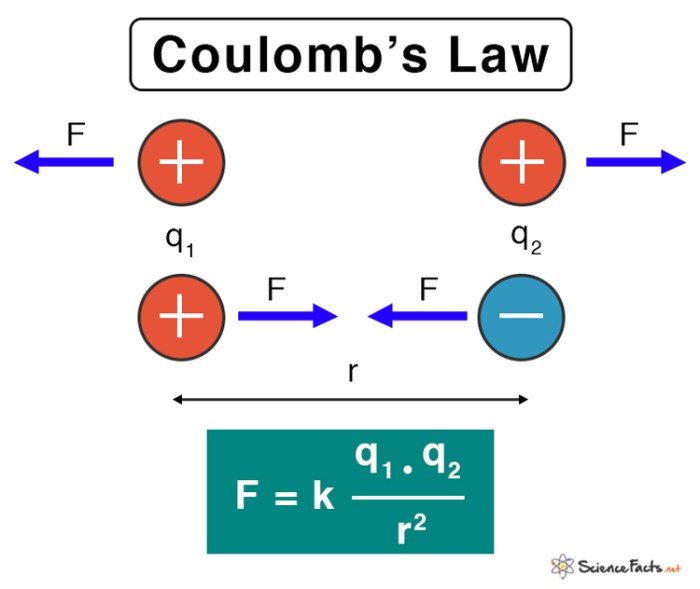
Coulomb’s law is a fundamental principle in electrostatics that describes the force of interaction between two charged objects. It has wide-ranging applications in various fields, including electronics, physics, and chemistry. Understanding how Coulomb’s law governs electrostatic interactions is crucial for developing technologies and understanding natural phenomena.
Applications in Electronics
Coulomb’s law is essential for understanding and designing electronic devices. Here are some examples:
- Capacitors: Coulomb’s law helps determine the capacitance of capacitors, which store electrical energy. Capacitance is directly proportional to the charge stored and inversely proportional to the voltage applied. Coulomb’s law explains how charges accumulate on the capacitor plates, creating an electric field.
- Transistors: Coulomb’s law plays a crucial role in the operation of transistors, which are fundamental building blocks of modern electronics. The flow of current in a transistor is controlled by the electric field created by charges on the gate, which is determined by Coulomb’s law.
- Integrated Circuits (ICs): Coulomb’s law governs the interactions between charges within ICs, determining the flow of current and the performance of circuits. Understanding these interactions is essential for designing and manufacturing ICs.
Applications in Physics
Coulomb’s law is a cornerstone of electromagnetism and has numerous applications in physics:
- Electrostatic Forces: Coulomb’s law directly quantifies the electrostatic force between charged particles. This force can be attractive or repulsive, depending on the signs of the charges.
- Electric Fields: Coulomb’s law is used to calculate the electric field created by a point charge or a distribution of charges. The electric field is a vector quantity that describes the force that a test charge would experience at a given point in space.
- Electrostatic Potential: Coulomb’s law can be used to calculate the electrostatic potential, which is a scalar quantity that describes the potential energy per unit charge at a given point in space. The potential is related to the electric field and provides information about the work done by the electric force.
Applications in Different Industries, What is k in coulomb’s law
| Industry | Applications of Coulomb’s Law | Examples |
|---|---|---|
| Electronics | Capacitor design, transistor operation, integrated circuit fabrication | Designing capacitors for energy storage in smartphones, developing high-speed transistors for computer processors, optimizing the layout of transistors in integrated circuits |
| Physics | Understanding electrostatic forces, calculating electric fields, determining electrostatic potential | Studying the interactions between charged particles in atomic and molecular systems, analyzing the behavior of charged particles in electric fields, developing new materials with specific electrical properties |
| Chemistry | Predicting the behavior of ionic compounds, understanding the interactions between molecules, designing new materials with desired properties | Predicting the solubility of ionic compounds in different solvents, studying the interactions between molecules in solution, developing new catalysts for chemical reactions |
| Biology | Understanding the interactions between charged molecules in biological systems, developing new drugs and therapies | Studying the interactions between DNA molecules, analyzing the behavior of proteins in cells, developing new drugs that target specific proteins |
Limitations of Coulomb’s Law: What Is K In Coulomb’s Law

Coulomb’s law is a fundamental principle in electrostatics, providing a simple and elegant description of the force between stationary charged particles. However, it’s important to recognize that this law has certain limitations, and its applicability can be restricted in specific scenarios. This section delves into the limitations of Coulomb’s law, exploring the conditions under which it might not accurately predict the force between charges.
Conditions Where Coulomb’s Law May Not Be Accurate
Coulomb’s law is a powerful tool for understanding electrostatic interactions, but it has limitations in certain situations. It’s crucial to understand these limitations to apply the law effectively and interpret the results correctly.
- For charges in motion: Coulomb’s law is strictly valid for stationary charges. When charges are in motion, their interaction is influenced by magnetic fields, which are not considered in Coulomb’s law. This means the force between moving charges will be different from the force predicted by Coulomb’s law alone.
- For charges in close proximity: Coulomb’s law assumes that the charges are point charges, meaning they have no size or structure. This assumption breaks down when charges are very close to each other. At very small distances, the internal structure of the charges and quantum effects become significant, and Coulomb’s law may not accurately describe the interaction.
- For charges in a medium: Coulomb’s law applies to charges in a vacuum. When charges are immersed in a dielectric medium, such as water or oil, the medium’s polarizability affects the interaction between the charges. The force between charges in a medium is typically weaker than in a vacuum due to the screening effect of the medium’s polarizable molecules.
Factors Affecting the Accuracy of Coulomb’s Law
Several factors can influence the accuracy of Coulomb’s law in describing the force between charges.
- Distance between charges: The accuracy of Coulomb’s law depends on the distance between the charges. At very small distances, quantum effects become significant, and Coulomb’s law may not be accurate.
- Presence of other charges: The presence of other charges near the interacting charges can affect the force between them. This is because the electric field created by each charge influences the other charges, leading to a more complex interaction than predicted by Coulomb’s law alone.
- Shape and size of the charges: Coulomb’s law assumes point charges, which have no size or structure. However, real charges have a finite size and shape, which can affect the force between them, especially at short distances.
Ending Remarks

Understanding Coulomb’s Law and the significance of ‘k’ is essential for grasping the fundamental principles of electromagnetism. From everyday phenomena like static electricity to complex technological applications like microchips, Coulomb’s Law provides a framework for comprehending how charges interact and shape the world around us. As we delve deeper into the world of electromagnetism, we discover that ‘k’ is not just a simple constant, but a gateway to a vast and intricate universe of electrical forces.
Clarifying Questions
Why is ‘k’ called Coulomb’s constant?
It’s named after Charles-Augustin de Coulomb, the French physicist who formulated the law describing the force between charges.
What is the numerical value of ‘k’?
The value of ‘k’ is approximately 8.98755 × 10⁹ N⋅m²/C² in the SI system.
How does ‘k’ affect the force between charges?
A larger value of ‘k’ implies a stronger force between the charges, while a smaller value indicates a weaker force.
Are there different values of ‘k’ for different units?
Yes, the value of ‘k’ depends on the units used for charge, distance, and force. For example, in the Gaussian system, the value of ‘k’ is 1.





