
What is the Third Law of Thermodynamics sets the stage for this enthralling narrative, offering readers a glimpse into a story that is rich in detail and brimming with originality from the outset. The Third Law of Thermodynamics, one of the fundamental laws governing the universe, delves into the intriguing world of absolute zero and its implications. This law dictates that as a system approaches absolute zero, its entropy, or the degree of disorder within it, approaches a constant value. This seemingly simple concept has profound implications for our understanding of the universe, impacting everything from the behavior of matter at extremely low temperatures to the ultimate fate of the cosmos.
This law is essential for understanding the behavior of matter at extremely low temperatures, a realm where quantum mechanics reigns supreme. It helps us understand the properties of materials at these temperatures, paving the way for groundbreaking technological advancements in fields like superconductivity and quantum computing. The Third Law of Thermodynamics also has implications for our understanding of the universe as a whole, shedding light on its ultimate fate and the concept of heat death.
Introduction to Thermodynamics
Thermodynamics is a branch of physics that deals with heat and its relationship to other forms of energy. It is a fundamental science that has wide-ranging applications in various fields, including engineering, chemistry, and biology.
Thermodynamics is concerned with the study of energy transfer and its transformations. It provides a framework for understanding how energy flows in systems and how it can be used to perform work.
Fundamental Concepts of Thermodynamics, What is the third law of thermodynamics
Thermodynamics is built upon several fundamental concepts that form the basis for understanding energy and its transformations. These concepts include:
- System: A system is a defined region of space that is being studied. It can be anything from a single atom to an entire planet. Systems can be open, closed, or isolated, depending on their ability to exchange energy and matter with their surroundings.
- Surroundings: The surroundings are everything outside the system that can interact with it. The surroundings can provide energy to the system, or the system can transfer energy to the surroundings.
- State: The state of a system is defined by its properties, such as temperature, pressure, and volume. The state of a system can change as it interacts with its surroundings.
- Energy: Energy is the ability to do work. In thermodynamics, energy can exist in various forms, including heat, mechanical energy, chemical energy, and electrical energy.
- Heat: Heat is the transfer of thermal energy between objects at different temperatures. Heat always flows from a hotter object to a colder object.
- Work: Work is the transfer of energy from one system to another by a force acting over a distance. In thermodynamics, work can be done by a system on its surroundings, or by the surroundings on the system.
Importance of the Three Laws of Thermodynamics
The three laws of thermodynamics are fundamental principles that govern the behavior of energy and its transformations. They provide a framework for understanding how energy flows in systems and how it can be used to perform work. The three laws of thermodynamics are:
- First Law of Thermodynamics: The first law of thermodynamics states that energy cannot be created or destroyed, only transferred or transformed from one form to another. This law is also known as the law of conservation of energy.
- Second Law of Thermodynamics: The second law of thermodynamics states that the total entropy of an isolated system can never decrease over time. Entropy is a measure of the disorder or randomness of a system. The second law implies that the universe is constantly becoming more disordered.
- Third Law of Thermodynamics: The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches absolute zero. Absolute zero is the lowest possible temperature, at which all molecular motion ceases. The third law implies that it is impossible to reach absolute zero.
The Third Law of Thermodynamics

The Third Law of Thermodynamics is a fundamental principle that governs the behavior of matter at extremely low temperatures. It describes the behavior of entropy, a measure of disorder or randomness in a system, as the temperature approaches absolute zero.
Absolute Zero and Its Implications
Absolute zero, defined as 0 Kelvin (-273.15 °C or -459.67 °F), represents the theoretical point at which all molecular motion ceases. At this temperature, a system reaches its lowest possible energy state, and its entropy attains a minimum value.
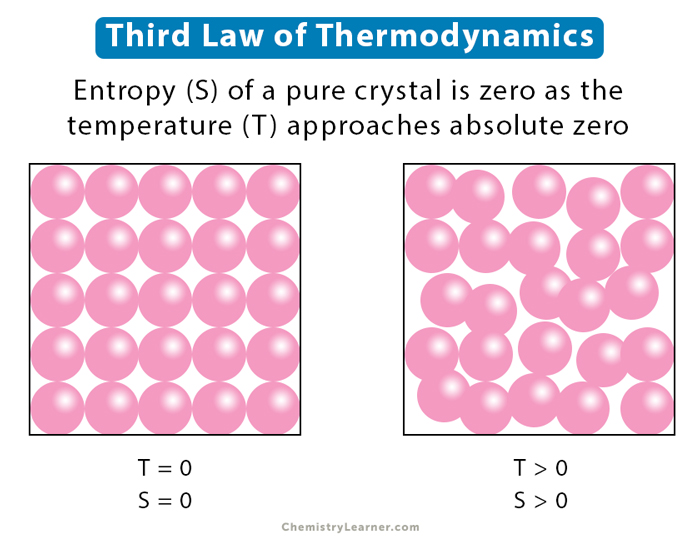
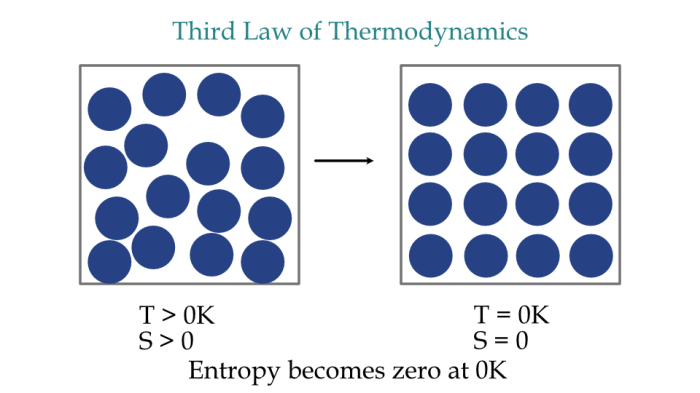
The Third Law of Thermodynamics states that the entropy of a perfect crystal at absolute zero is zero.
This implies that at absolute zero, a system is perfectly ordered, with no thermal energy to cause any motion or randomness. The implications of this law are far-reaching:
- Perfect Order: At absolute zero, a system is perfectly ordered, with no thermal energy to cause any motion or randomness.
- Minimum Entropy: The entropy of a system reaches its minimum value at absolute zero.
- Theoretical Limit: Absolute zero represents a theoretical limit for the temperature of a system.
- Cryogenics and Quantum Phenomena: The Third Law has significant implications for cryogenics, the study of extremely low temperatures, and for understanding quantum phenomena.
The Unattainability of Absolute Zero
While absolute zero is a theoretical concept, it is practically impossible to reach. This is because of the following reasons:
- Quantum Fluctuations: Even at extremely low temperatures, quantum fluctuations in energy prevent a system from reaching absolute zero.
- Heat Transfer: It is impossible to completely isolate a system from its surroundings, and even the smallest amount of heat transfer can prevent it from reaching absolute zero.
- Cooling Techniques: Cooling techniques, such as adiabatic demagnetization, can achieve temperatures extremely close to absolute zero, but they cannot reach absolute zero itself.
The unattainability of absolute zero has practical implications for various fields:
- Cryogenics: Cryogenic systems are designed to operate at extremely low temperatures, but they cannot reach absolute zero. This has implications for the design and performance of cryogenic equipment.
- Quantum Computing: Quantum computers operate at extremely low temperatures to minimize thermal noise and improve coherence. The unattainability of absolute zero is a factor in the design and development of these computers.
- Material Science: The Third Law has implications for the study of materials at low temperatures, including superconductivity and superfluidity.
Conclusion: What Is The Third Law Of Thermodynamics
The Third Law of Thermodynamics, though seemingly simple, holds the key to unlocking profound mysteries about the universe. It guides us through the intricate dance of energy and entropy, revealing the secrets of matter at its most fundamental level. This law not only serves as a cornerstone of modern physics but also opens up avenues for technological advancements and philosophical explorations into the very essence of existence.
Essential FAQs
What are some real-world applications of the Third Law of Thermodynamics?
The Third Law has applications in various fields, including:
- Chemistry: Understanding chemical reactions and equilibrium at low temperatures.
- Materials Science: Developing new materials with unique properties at low temperatures, such as superconductors.
- Engineering: Designing efficient refrigeration and cryogenic systems.
Can we ever reach absolute zero?
While the Third Law states that absolute zero is unattainable, we can get incredibly close to it in laboratory settings. This has led to the development of cryogenics, the study of extremely low temperatures.
What is the difference between the Third Law and the other laws of thermodynamics?
The First Law of Thermodynamics deals with the conservation of energy, while the Second Law deals with the increase in entropy in isolated systems. The Third Law focuses on the behavior of entropy at absolute zero.




